Maleic acid is converted to fumaric acid because fumaric acid is more stable. Chemists are interested in why maleic acid is converted to fumaric acid through isomerization. In this process, maleic acid molecules rearrange themselves, like puzzle pieces finding better positions, ultimately transforming into fumaric acid. When maleic acid is converted to fumaric acid, atoms move to achieve a more comfortable and stable arrangement. This transformation occurs in a chemical reaction, which is crucial for the production of fumaric acid for various applications. The process of maleic acid converting to fumaric acid highlights how chemistry influences the products we use every day.
Key Takeaways
- Maleic acid is converted to fumaric acid because fumaric acid is more stable. This makes fumaric acid a better choice for many applications.
- Isomerization is the process by which maleic acid changes its form to convert into fumaric acid. Heating or a catalyst usually triggers this transformation. The newly formed molecule is more stable.
- Fumaric acid has some unique properties. It is not easily soluble and has a higher melting point than maleic acid. This makes it valuable in the food, pharmaceutical, and industrial sectors.
- The rate of conversion of maleic acid to fumaric acid increases with increasing temperature. This indicates that heat is crucial to this conversion process.
- Fumaric acid is safe for consumption. It is used in food, animal feed, and pharmaceuticals, demonstrating its important role in daily life.
Isomerization: How maleic acid is converted to fumaric acid

Cis-Trans Isomerism Explained
Chemists define isomerism as the phenomenon where molecules with the same atomic structure exhibit different conformations. For maleic acid and fumaric acid, this phenomenon is called cis-trans isomerism. Isomerization of maleic acid means that the positions of certain groups around the double bond have changed. In maleic acid, the two carboxyl groups are located on the same side of the double bond, while in fumaric acid, the two carboxyl groups are located on opposite sides.
This conformational change results in drastically different properties. The groups in maleic acid (cis isomer) are closely packed, while those in fumaric acid (trans isomer) have more space between them. This makes fumaric acid more stable. Maleic acid is converted to fumaric acid because the molecule tends to acquire a more comfortable and stable conformation. Scientists call this phenomenon chemical isomerization.
There are many ways in which maleic acid isomerizes to fumaric acid. Sometimes, heating or light can initiate the reaction. Chemists can also use catalysts, such as mineral acids or peroxides, to accelerate the reaction process. Fumaric acid is needed in many fields, so chemists have developed special conversion methods. Sometimes, the isomerization of maleic acid can be completed with only water and heat, without the need for a catalyst.
The main steps of this process are as follows:
- Maleic acid is converted to fumaric acid under heating or with the aid of a catalyst.
- Common catalysts include mineral acids, peroxides, and thiourea.
- Fumaric acid can be purified by water crystallization.
Fumaric acid can also be prepared in one step using hot water without the use of a catalyst. Studies have shown that the isomerization of maleic acid to fumaric acid can occur without a catalyst.
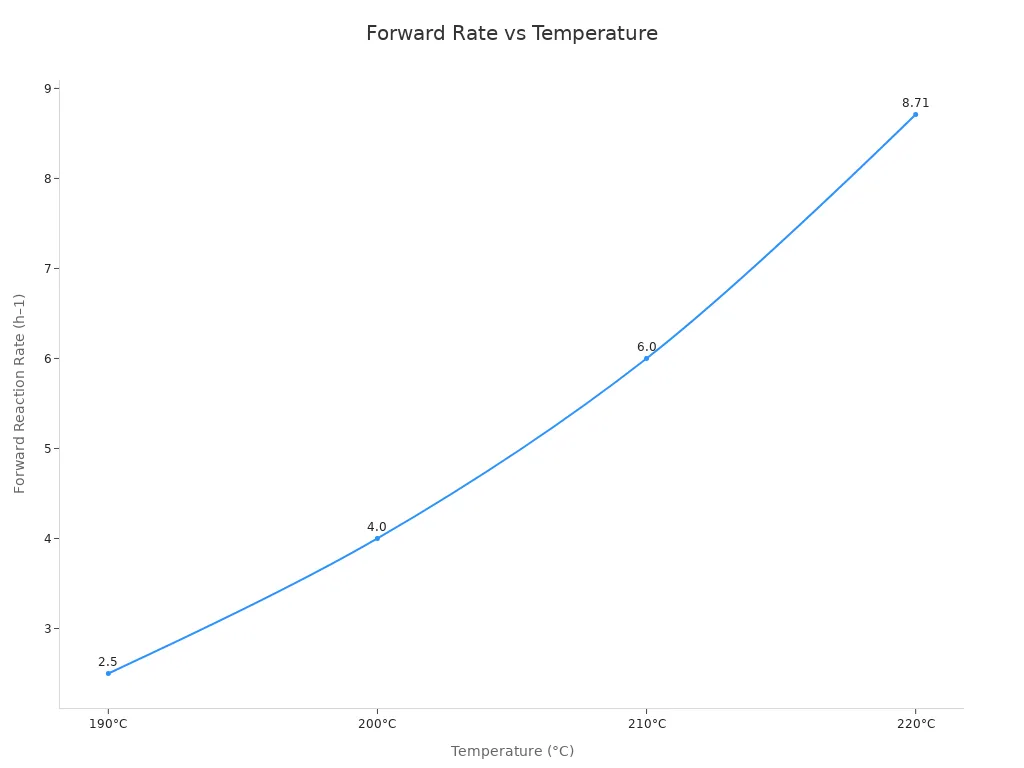
Experiments also support this conclusion. As the temperature increases, the forward rate constant for the isomerization of maleic acid to fumaric acid increases significantly. This means that higher temperatures accelerate the isomerization reaction.
| Temperature (°C) | Forward Reaction Rate Constant (h–1) | Reverse Reaction Rate Constant (h–1) |
|---|---|---|
| 190 | 2.5 | < 0.32 |
| 200 | 4.0 | < 0.32 |
| 210 | 6.0 | < 0.32 |
| 220 | 8.71 | < 0.32 |
Initially, the amount of fumaric acid increases over time. After an hour, its amount may decrease because some fumaric acid will revert to maleic acid or malic acid. This indicates that the reaction can proceed in both directions, depending on the reaction conditions.
Structural Differences between Maleic Acid and Fumaric Acid
Maleic acid and fumaric acid appear structurally almost identical, but their differences are crucial. Both contain the same atoms, but their groups are linked differently. Maleic acid is a cis isomer, so its carboxyl group is located on the same side of the molecule. Fumaric acid is a trans isomer, with its carboxyl group located on both sides of the molecule.
This subtle difference results in drastically different properties. Maleic acid is more polar and more soluble in water. Fumaric acid is less polar and less soluble. Maleic acid has a lower melting point. Fumaric acid has a higher melting point. Isomerization of maleic acid to fumaric acid makes the molecule more stable.
| Isomer Type | Structure | Stability |
|---|---|---|
| Cis | Maleic acid (cis-butenedioic acid) | Usually less stable because of crowding |
| Trans | Fumaric acid (trans-butenedioic acid) | More stable because there is less crowding |
| Property | Maleic Acid | Fumaric Acid |
|---|---|---|
| Dipole Moment | Polar | Nonpolar |
| Solubility | High | Low |
| Melting Point | Lower | Higher |
| Stability | Less stable | More stable |
| pKa Value | Lower | Higher |
| Reactivity | More reactive | Less reactive |
| Property | Maleic Acid (Cis) | Fumaric Acid (Trans) |
|---|---|---|
| Geometry | Groups on same side, more strain | Groups on opposite sides, less strain |
| Stability | Lower | Higher |
| Melting Point | Lower | Higher |
| Solubility | Higher in water | Lower in water |
Isomerization converts maleic acid to fumaric acid, transforming the molecule from an unstable and highly reactive configuration to a more stable and less reactive one. This chemical isomerization is crucial for the production of products requiring long-term storage. The isomerization reaction of maleic acid is a key reaction in many industries, especially those where fumaric acid is needed to exert its specific properties.
Conditions for Maleic Acid Isomerization
Heating and Acid Catalysts
Heating and acid catalysts are essential for the conversion of maleic acid to fumaric acid. This reaction requires energy to initiate molecular motion and undergo a configurational change. Scientists utilize heating to accelerate this process. Acid catalysts such as phosphoric acid facilitate the configurational change of maleic acid. This allows maleic acid to transform from the cis configuration to the more stable trans configuration.
The reaction is most effective under appropriate heating and acid concentrations. The table below shows how temperature affects the reaction rate constant:
| Temperature (°C) | Reaction Rate Constant (h–1) |
|---|---|
| 190 | 2.5 |
| 220 | 8.71 |
Higher temperatures accelerate the conversion. The optimal acid concentration is between 100 and 130 g dm⁻³. Scientists also test the carbon-to-nitrogen ratio to increase the yield of fumaric acid. Suitable conditions facilitate the production of large quantities of pure fumaric acid.
Hydrothermal and Photochemical Methods
The hydrothermal method utilizes hot water to convert maleic acid. This method requires no catalyst. The conversion is completed in one step, with a fumaric acid yield of up to 92%. The process works well at high temperatures and is typically completed in about one hour. The key steps of this method are listed in the table below:
| Aspect | Details / Results |
|---|---|
| Reaction type | Batch noncatalytic isomerization |
| Yield of fumaric acid | Up to 92% |
| Reaction conditions | Hydrothermal, one-step, catalyst-free |
| Product characterization | X-ray diffraction and FTIR confirm fumaric acid |
| Influencing factors | Temperature, time, initial maleic acid concentration |
Photochemical methods utilize light energy to initiate the conversion process. When maleic acid absorbs light, the energy promotes molecular motion, ultimately forming fumaric acid. Both hydrothermal and photochemical methods are effective ways to prepare fumaric acid from maleic acid. These methods help various industries choose the optimal process based on their specific needs.
Advantages of Fumaric Acid
Thermodynamic Stability
Chemists favor fumaric acid because it is more stable than maleic acid. When maleic acid is converted to fumaric acid, atoms move. Fumaric acid has a trans configuration, while maleic acid has a cis configuration. The trans configuration allows molecules to pack together closely, thus strengthening intermolecular bonds. The cis configuration of maleic acid causes the groups in the molecules to be too close together, leading to molecular crowding and weakened bonds. Converting maleic acid to fumaric acid increases the yield of fumaric acid and makes it more stable.
| Acid Type | Configuration | Stability Reason |
|---|---|---|
| Fumaric Acid | Trans | Allows for tight packing and stronger intermolecular bonds, leading to higher stability. |
| Maleic Acid | Cis | Results in weaker bonds and increased molecular strain, making it less stable. |
Isomerization facilitates the production of more fumaric acid. Scientists utilize this change to obtain a stable product. The amount of fumaric acid depends on the reaction conditions. If the temperature and catalyst are suitable, the yield increases.
Energy Driven
The conversion of maleic acid to fumaric acid requires energy. Heat, light, or a catalyst can initiate the reaction. Because fumaric acid is more stable, the reaction produces more fumaric acid. As the reaction proceeds, more fumaric acid is produced. Studies have shown that fumarate enzymes facilitate this change and can remain stable for at least 10 nanoseconds. This explains why fumaric acid is more stable.
Scientists have observed that the reaction produces more fumaric acid because the molecules tend to be in a lower energy state.
The reaction continues until the maximum amount of fumaric acid is reached. When equilibrium is reached, the reaction stops. Chemists monitor the amount of fumaric acid to ensure it is sufficient. The conversion of maleic acid to fumaric acid is important in many industries. The reaction produces a stable compound that can be used in food, pharmaceuticals, and other products.
Uses of Fumaric Acid in Industry and Applications

Food and Beverage Applications
NORBIDAR is a leading producer of fumaric acid in the food and beverage industry. Converting maleic acid to fumaric acid yields a product with high stability and high yield. Food manufacturers use fumaric acid to improve the taste of food and maintain freshness. This reaction produces a more lasting acidity than other acids. Fumaric acid helps maintain the appropriate acidity in cheese and yogurt and also prevents powdered beverages from clumping. The table below lists how fumaric acid is used in different foods:
| Application Area | Description |
|---|---|
| Beverages | Makes juices and soft drinks more tart and controls acidity. |
| Baked Goods | Makes dough stronger and keeps baked goods fresh longer. |
| Confectionery | Gives candies and gummies a sour taste. |
| Dairy Products | Balances acidity and makes cheese and yogurt smoother. |
| Processed Foods | Improves flavor and keeps foods fresh. |
Animal Nutrition and Feed Conversion Rate
Farmers use fumaric acid to help animals obtain better nutrition. Converting maleic acid to fumaric acid yields more of this beneficial feed component. Fumaric acid helps chickens, pigs, and fish maintain healthy stomachs. It helps animals absorb nutrients and grow faster. Studies have shown that this reaction can reduce methane emissions in cattle and sheep. The table below shows how fumaric acid helps different animals:
| Livestock Segment | Uses in Feed | Observed Benefits |
|---|---|---|
| Poultry | Makes stomachs healthier, stops bad bacteria | Safer food |
| Swine | Makes feed work better, helps piglets grow | Faster growth |
| Ruminants | Helps with digestion, makes animals eat more | Healthier animals |
| Aquaculture | Makes water cleaner, helps fish grow | Healthier fish |
Personal Care and Pharmaceuticals
Fumaric acid is also used in skincare and pharmaceuticals. Converting maleic acid to fumaric acid yields a high-purity, high-yield product. Skincare products utilize fumaric acid to balance skin pH and gently cleanse the skin. In haircare, it makes hair smoother and shinier. Pharmaceutical companies use fumaric acid to treat psoriasis and multiple sclerosis. Dimethyl fumarate, produced by this reaction, helps treat certain diseases. Doctors use ferrous fumarate as an iron supplement to treat anemia.
Industrial Materials and Resins
Plants use fumaric acid to produce strong and durable materials. Converting maleic acid to fumaric acid yields a wide range of products used in resins and coatings. Fumaric acid enhances the strength of unsaturated polyester resins. These resins are used in automobiles, ships, and construction. Fumaric acid also speeds up paint drying and extends its lifespan. NORBIDAR offers high-quality fumaric acid suitable for the above applications, helping products maintain strength and being environmentally friendly.
The global market for fumaric acid is growing rapidly. Experts predict that its market size will exceed $1 billion by 2030. This highlights the importance of efficient and high-quality fumaric acid production.
Maleic acid is converted to fumaric acid because the newly formed molecule is more stable. Scientists study this reaction to explore how molecules tend towards equilibrium and stability. The conversion of maleic acid to fumaric acid has significant benefits for numerous industries, including food, pharmaceuticals, and materials manufacturing. NORBIDAR is a leading company in fumaric acid production. They employ new technologies and safe production methods, and are committed to ensuring that every step of the reaction is carried out safely and efficiently.
| Evidence Type | Details |
|---|---|
| Experience | Over 15 years in the production of fumaric acid. |
| Production Capacity | Capable of producing up to 30,000 tons of fumaric acid annually. |
| Commitment to Quality | Focus on high purity food-grade fumaric acid and adherence to safety certifications. |
| Innovative Manufacturing | Utilizes advanced technology and stringent production standards. |
| Environmental Commitment | Employs bio-fermentation and renewable raw materials to minimize environmental impact. |
FAQ
What is fumaric acid used for?
Fumaric acid has a wide range of uses. It’s added to food and beverages. It’s also used in animal feed, skincare products, and pharmaceuticals. Factories use it to produce high-strength products. Fumaric acid helps extend the shelf life of food and improves its taste. Many businesses need fumaric acid to achieve its specific benefits.
Why does maleic acid convert to fumaric acid?
This reaction occurs because fumaric acid is more stable. Molecules tend to remain stable. This reaction allows atoms to move to more stable positions. Scientists utilize this property to obtain more fumaric acid for various applications.
How does heating affect the fumaric acid formation reaction?
Heating provides energy for the reaction. When scientists heat the mixture, the reaction speeds up. More fumaric acid is produced in a shorter time. High temperatures help molecules change shape. This makes the reaction more efficient in producing fumaric acid.
Is fumaric acid edible?
Experts say that fumaric acid is safe to use in food. Food manufacturers use fumaric acid to enhance the sourness and freshness of food. The synthesis of fumaric acid follows strict rules. Food safety agencies conduct quality testing before people consume foods containing fumaric acid.
Can the synthesis of fumaric acid be carried out without a catalyst?
Yes, the reaction can be carried out without a catalyst. Hot water can initiate the reaction and produce fumaric acid. Scientists have found this method to be very effective. Even without the addition of other chemicals, the reaction can still produce large amounts of fumaric acid.