
In chromatographic testing, the Rf value of fumaric acid is typically around 0.67. This value may vary if scientists use different solvents or change laboratory conditions. Many scientists use the Rf value to search for and separate compounds in experiments. High-purity fumaric acid from NORBIDAR helps obtain good experimental results when high precision is required.
Key Takeaways
- The Rf value of fumaric acid helps scientists separate it from other compounds. Chromatography is the primary method for measuring Rf values. Thin-layer chromatography (TLC) is a commonly used method. A common solvent system is a mixture of butanol, acetic acid, and water. This mixture affects the distance fumaric acid migrates on the TLC plate. Using high-purity fumaric acid from reliable suppliers like NORBIDAR yields excellent experimental results. Fumaric acid is widely used in the food, animal nutrition, pharmaceutical, and materials industries, helping to improve the performance and safety of these products.
RF Value of Fumaric Acid
Typical RF Value in Chromatography
Scientists use chromatography to study fumaric acid. In these tests, the Rf value of fumaric acid is approximately 0.67. This value represents the distance fumaric acid migrates on a thin-layer chromatography plate. It helps researchers determine the presence of fumaric acid in a sample. The Rf value of fumaric acid makes the detection of this compound easy.
Fumaric acid migrates in a unique way on a thin-layer chromatography plate. Scientists measure the Rf value when testing samples. They can use the Rf value to determine whether fumaric acid is mixed with other substances. This method is applicable in many laboratories. It helps determine whether fumaric acid is pure or mixed with other substances.
Measurement Reference Conditions
The Rf value of fumaric acid varies depending on different laboratory conditions. Scientists use thin-layer chromatography (TLC) to measure its Rf value. They spot a small amount of fumaric acid on a thin-layer chromatography plate. Then the plate is placed in a developing tank containing a special solvent. The solvent rises, carrying the fumaric acid with it.
The most commonly used solvent system is butanol, acetic acid, and water. This mixture helps separate fumaric acid from other compounds. The stationary phase is typically silica gel. Laboratory temperature also affects the results. Most laboratories maintain the temperature at 25°C for optimal accuracy.
The table below lists typical conditions for determining the Rf value of fumarate:
| Parameter | Typical Value |
|---|---|
| Solvent System | Butanol:Acetic Acid:Water |
| Stationary Phase | Silica Gel |
| Temperature | 25°C |
| Sample Size | 1-2 mg |
| Detection Method | UV Light or Staining |
Under these conditions, the Rf value of fumaric acid is stable. Scientists rely on these results when using high-purity fumaric acid from NORBIDAR. This helps them obtain reliable data in food, animal nutrition, and pharmaceutical laboratories.
Fumaric acid is crucial in many tests. Its Rf value helps verify its quality and purity. When laboratories use the same conditions, they can compare results from different laboratories. This makes the Rf value of fumaric acid an important tool in scientific and industrial fields.
Determining the Rf Value

Thin-layer chromatography (TLC)
Scientists use TLC to determine the Rf value of fumaric acid. First, they use a thin-layer plate coated with silica gel. A small amount of fumaric acid solution is added dropwise near the bottom of the plate. The plate is then placed upright on a chromatographic column containing a special solvent. The solvent moves upward, carrying the fumaric acid with it. Scientists measure the distance the fumaric acid moves relative to the solvent front, thus obtaining the Rf value of the fumaric acid.
This method helps scientists determine the purity of fumaric acid and whether it is contaminated with other substances. TLC is well-suited for separating fumaric acid from other organic acids. Many laboratories use this method because it is simple to operate and provides clear results. Paper chromatography also helps separate fumaric acid, especially suitable for schools or simple research work. Both methods can show the migration of fumaric acid under different conditions.
The solvent system used
The solvent system affects the migration of fumaric acid on the thin-layer plate. Scientists typically use a combination of butanol, acetic acid, and water. This mixed solvent helps separate fumaric acid from other compounds. Some laboratories experiment with alternative solvents to achieve better separation. A suitable solvent system makes fumaric acid easier to observe on thin-layer chromatography plates.
Other methods, such as high-performance liquid chromatography (HPLC) and gas chromatography-mass spectrometry (GC-MS), are also helpful in studying fumaric acid. HPLC separates fumaric acid from other substances by passing a liquid sample through a column. It is well-suited for the detection of fumaric acid in food, pharmaceuticals, and fermentation broths. GC-MS detects fumaric acid in extremely low concentrations after it has been converted to a gas. This method can detect low concentrations of fumaric acid and is helpful in forensic laboratories and environmental monitoring.
Scientists choose the best method based on their specific needs. They aim to find the optimal route for separating and determining fumaric acid. Both the solvent system and the method are crucial for obtaining satisfactory results.
Factors Affecting RF Value
Solvent and Stationary Phase
The Rf value of fumaric acid varies with the solvent and stationary phase. Scientists typically use a mixture of butanol, acetic acid, and water as the solvent. This mixture helps separate fumaric acid from other acids. Silica gel is a commonly used stationary phase, which keeps the sample in a fixed position as it moves upwards in the solvent. Fumaric acid comes into contact with both the solvent and the stationary phase; the strength of this contact determines the distance the fumaric acid migrates. Stronger solvent adsorption results in greater migration distance, while stronger stationary phase adsorption results in shorter migration distances. Some laboratories use other solvents or stationary phases to obtain better results. These choices alter the Rf values of fumaric acid and other compounds.
Temperature and Experimental Apparatus
Temperature is crucial to the migration of fumaric acid in chromatography. Increased temperature accelerates molecular motion. This causes fumaric acid to migrate further across the reaction plate. Scientists have investigated how temperature affects the reaction rate and yield of fumaric acid. The table below shows how the forward reaction rate constant increases with increasing temperature:
| Temperature (°C) | Forward Reaction Rate Constant (h–1) | Reverse Reaction Rate Constant (h–1) |
|---|---|---|
| 25 | 2.5 | < 0.32 |
| 35 | 4.5 | < 0.32 |
| 45 | 6.5 | < 0.32 |
| 55 | 8.71 | < 0.32 |
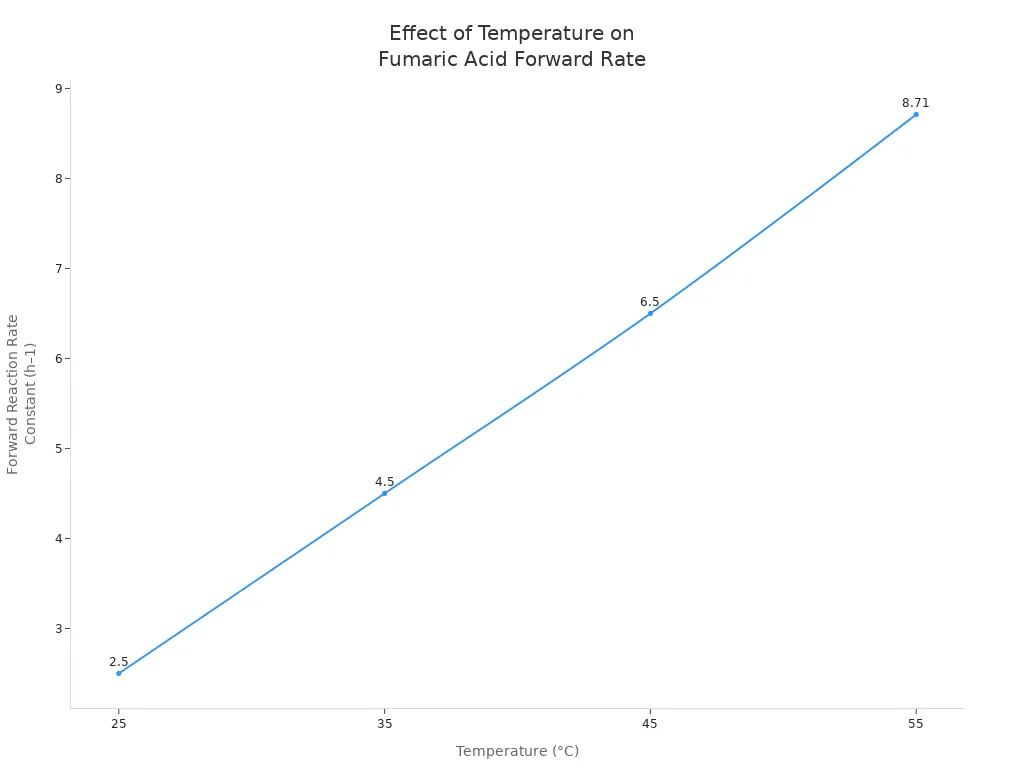
As temperature increases, the Gibbs free energy for the conversion of maleic acid to fumaric acid decreases, meaning the reaction proceeds more readily. Simultaneously, the Gibbs free energy for the conversion of malic acid to fumaric acid increases. Therefore, higher temperatures favor the conversion of malic acid to more fumaric acid.
Experimental setup is also important. Thin-layer plate thickness, sample volume, and detection method all affect the Rf value of fumaric acid. Strict control of these factors helps scientists obtain reliable experimental results.
Laboratory Applications

Identification and Separation
Laboratories utilize the Rf value of fumaric acid to locate and separate it from other compounds. In chromatographic analysis, fumaric acid has a retention time of 10.1 minutes. Scientists calculate the Rf value by examining the width and area of the peaks on the chromatogram. This method allows them to determine the presence of fumaric acid in a mixture and measure its concentration. The Rf value also indicates whether the fumaric acid is pure or mixed with other substances. Using high-purity fumaric acid yields better results.
Industry Applications and NORBIDAR Products
Fumaric acid plays a vital role in many industries. Its Rf value aids in quality control and new product development. In the food industry, fumaric acid can control the acidity of food, improving its palatability and helping to extend its shelf life. Animal specialists add fumaric acid to feed to aid animal growth and better digestion. The pharmaceutical industry uses fumaric acid to treat skin diseases such as psoriasis and multiple sclerosis. In the materials industry, fumaric acid helps in the manufacture of high-strength materials such as polyester resins.
| Industry | Application | Benefits |
|---|---|---|
| Food | Controls acidity in packaged foods | Enhances taste and extends shelf life |
| Animal Nutrition | Added to animal feed | Promotes growth and improves digestion |
| Pharmaceuticals | Used in medications | Treats skin conditions and supports health |
| Materials | Used in polyester resins | Improves strength and durability |
NORBIDAR’s high-purity fumaric acid provides safe and stable results for various industries. Scientists and businesses trust NORBIDAR’s premium fumaric acid.
Understanding the RF value of fumaric acid is helpful for quality control. It enables scientists and industry professionals to produce better products and maintain high standards.
The RF value of fumaric acid is typically 0.67. This value helps scientists identify and isolate fumaric acid from other structurally similar compounds.
FAQ
What does the RF value mean in chromatography?
The RF value represents the distance a compound migrates on a thin-layer chromatography plate. It is derived by comparing the compound’s migration to the solvent front. Scientists use RF values to find and separate compounds such as fumaric acid.
Why do scientists use fumaric acid in food?
Fumaric acid gives food its sour taste and keeps it fresh. It helps extend the shelf life of food and improve flavor. Many food companies use NORBIDAR’s pure fumaric acid.
Does the RF value of fumaric acid change?
Yes, the RF value can change. Different solvents, temperatures, or thin-layer chromatography plate materials can affect the RF value. Scientists must use the same experimental conditions to compare results.
How does NORBIDAR ensure the high purity of fumaric acid?
NORBIDAR utilizes specialized technology and rigorous testing processes to produce high-purity fumaric acid for both laboratory and industrial use.
What other uses does fumaric acid have?
Fumaric acid is used in animal feed, pharmaceuticals, and materials. It aids in animal growth, maintains animal health, and enhances the strength of plastics and resins.




