Fumarase converts fumaric acidto L-malate. This reaction is crucial for energy metabolism and cellular repair in the human body. The process occurs in both mitochondria and the cell nucleus. This conversion is bidirectional, allowing cells to adjust as needed.
| Evidence Description | Impact on Cellular Metabolism |
|---|---|
| Fumarase helps change fumaric acid to L-malic acid in the TCA cycle. | This change is needed for energy and other cell processes. |
| The change can go both ways. | Cells can change energy and repair work because of this. |
Key Takeaways
- Fumarase converts fumarate to L-malate. L-malate helps cells produce energy.
- This enzyme, found in mitochondria and the cell nucleus, is crucial for the tricarboxylic acid cycle.
- L-malate is essential for cellular metabolism, helping to maintain cellular energy stability and aiding in cellular repair.
- Fumarate has a dual catalytic function, enabling cells to adjust according to their energy and repair needs.
- Fumarate has wide applications in the food, pharmaceutical, and industrial sectors, demonstrating its significant importance in both biology and technology.
Fumarase Activity and Fumaric Acid
Fumaric Acid Overview
Fumaric acid is found in many plants and fungi. Scientists initially extracted fumaric acid from plants of the genus ‘Corydalis’. It is also found in rice, sugarcane, wine, leaves, and mushrooms. Microorganisms produce fumaric acid in nature through fermentation. In the human body, the concentration of fumaric acid in blood plasma is approximately 293 μM. Fumaric acid is a component of the tricarboxylic acid cycle, which helps cells obtain energy.
NORBIDAR produces fumaric acid for various industries. Their products can improve the taste of food and maintain its freshness. It is used as an acidifier and pH adjuster in beverages and baked goods. In animal nutrition, fumaric acid helps animals digest food and grow. It can reduce harmful bacteria and improve feed utilization. Pharmaceutical companies use fumaric acid in drugs and coatings.
Fumaric acid is important in both nature and industry. It is used in food, animal nutrition, personal care, pharmaceuticals, and manufacturing.
| Sector | Application Description |
|---|---|
| Food | Makes food taste better, keeps food fresh, controls acid and pH |
| Animal Nutrition | Helps digestion, supports growth, lowers bad bacteria |
| Pharmaceuticals | Used in medicines, protects cells and nerves |
| Personal Care | Conditions skin, exfoliates, brightens skin |
| Industrial Uses | Makes resins, plastics, coatings, improves strength and resistance |
Fumarase Function
Fumarate enzyme converts fumaric acid to L-malate. This enzyme functions in the mitochondria and nucleus of cells. Fumarase is the starting material for this reaction. Fumarase combines with water to produce L-malate.
Fumarate enzyme is essential for metabolism. Without fumarate enzyme, cells cannot convert fumaric acid to L-malate. This enzyme makes the reaction proceed quickly and smoothly. Fumarase helps cells maintain health and produce energy.
- Fumarate enzyme uses fumaric acid as a starting material.
- Fumarate enzyme produces L-malate, which cells need to provide energy.
- Fumarase maintains the balance of the tricarboxylic acid cycle.
Fumarate enzymes link the applications of fumaric acid in biology and industry. This reaction demonstrates how fumaric acid promotes life and technological advancements.
L-malic Acid: Product of Fumarase Action

Identity of L-malic Acid
L-malate is a naturally occurring compound found in living organisms and is essential for cellular energy production. Scientists know that L-malate is a key component of the tricarboxylic acid cycle. Fumarase and L-malate work synergistically in this cycle to help cells obtain energy. Fumarase converts fumarate to L-malate, allowing the cycle to continue.
The laboratory uses a special reaction to detect and measure L-malate. In this test, L-malate reacts with NAD+ under alkaline conditions. This reaction converts L-malate to oxaloacetate and generates NADH. Scientists measure the absorbance at a wavelength of 366 nm to determine the L-malate content in the sample.
| Step | Description |
|---|---|
| 1 | First plate reading without enzyme to set a baseline. |
| 2 | Enzyme is added and left for 21 minutes. |
| 3 | Final reading checks optical density. |
| 4 | Mean, standard deviation, and %CV are calculated with SoftMax Pro Software. |
| 5 | A standard curve is made to find L-malic acid levels. |
| 6 | L-malic acid in wine samples is found using the curve. |
Scientists use these steps to find and measure L-malic acid in many samples like food and drinks.
Fumarase and L-malic acid are important in science and industry. They help researchers learn how cells use energy. They also help check the quality of products like wine.
Biological Role
L-malic acid plays multiple roles in cells. Fumarase and L-malic acid are essential for the tricarboxylic acid cycle (TCA cycle). This cycle helps cells produce energy and maintain health.
- L-malic acid is a key component of the TCA cycle and energy metabolism.
- It acts as a signaling molecule, influencing cellular processes.
- Changes in L-malic acid levels are associated with certain diseases.
- L-malic acid is essential for mitochondrial energy supply.
- It acts as a signaling molecule, helping to control cellular function.
- L-malic acid is an intermediate product of the TCA cycle.
- It is essential for cellular metabolism and energy.
Fumarase and L-malate help organisms produce energy. L-malate is used as fuel and helps maintain cellular homeostasis. Fumarase converts L-malate into pyruvate and carbon dioxide. This produces NADPH, which is essential for energy and cellular health.
Fumarate and L-malate are crucial for energy, cell repair, and signal transduction. They help cells regulate and maintain health.
Fumarate and L-malate also affect how cells cope with stress and disease. If L-malate levels change, cells may experience problems with energy or repair. Scientists study fumarate and L-malate to understand disease and find new treatments.
Fumarase-Catalyzed Conversion

Water Addition Step
Fumarase is crucial for cellular metabolism. This enzyme converts fumarate to L-malate in a process achieved by adding a single water molecule. The reaction occurs in both mitochondria and the cytoplasm. Fumarase is most active at pH 7. Scientists have observed a conversion rate of approximately 80% in batch mode. Excessive accumulation of L-malate slows the reaction rate. Cells utilize this cycle to obtain energy from food.
Fumarase helps cells keep making energy. The enzyme acts fast and keeps the cell balanced.
Simple Reaction Process
Fumarate converts fumarate to L-malate in a simple manner. The enzyme has a strong affinity for fumarate. The affinity of fumarate in the cytoplasm for fumarate is much higher than for L-malate, binding to fumarate at a probability 17 times greater. This means the enzyme is more active. When cells produce more fumarate, they acquire more L-malate. There is no excess fumarate remaining. Nuclear magnetic resonance (NMR) studies have shown that fumarate is not easily reversed intracellularly.
| Evidence Type | Description |
|---|---|
| Enzyme Affinity | Cytosolic fumarase binds fumaric acid 17 times better than L-malic acid. |
| Enzyme Behavior | More fumarase makes more L-malic acid without extra fumaric acid. |
| NMR Studies | Fumarase does not switch between fumaric acid and L-malic acid in living cells. |
The reaction begins with fumarate binding to fumarate. The enzyme adds water to produce L-malate. The reaction stops when a certain amount of L-malate is reached. Cells require fumarate to maintain the normal functioning of the tricarboxylic acid cycle. Fumarase ensures adequate energy production. This enzyme helps cells maintain health and growth.
Fumarase and Its Role in Metabolism
Energy Production
Fumarate is essential for cellular energy production. This enzyme converts fumarate to L-malate in the tricarboxylic acid cycle (TCA cycle). The TCA cycle, also known as the Krebs cycle, occurs in the mitochondria. Cells require fumarate enzymes to maintain the proper functioning of the TCA cycle.
Fumarate enzymes play a role far beyond providing energy. This enzyme is also involved in amino acid metabolism and the urea cycle. These steps help cells synthesize proteins and remove waste products. Fumarases connect different metabolic pathways, ensuring cells receive the nutrients they need.
If fumarate enzymes malfunction, cells cannot produce enough energy. Fumarate accumulates, preventing pyruvate from entering the mitochondria. This slows the TCA cycle, leading to reduced ATP production. Cells may switch to glycolysis, but glycolysis also produces less energy. Some cells will alter their energy utilization to survive.
| Key Findings | Description |
|---|---|
| Reduced Pyruvate Import | Cells with less fumarase have less pyruvate in mitochondria. |
| TCA Cycle Activity | Low fumarase slows the tca cycle and lowers ATP. |
| Shift to Glycolysis | Cells may switch to glycolysis for energy. |
TCA Cycle Significance
The tricarboxylic acid cycle (TCA cycle) is essential for life. Fumarase maintains this cycle. This enzyme converts fumarate to L-malate. This step is necessary for the proper functioning of the TCA cycle. Without fumarate, the TCA cycle slows down, and cells cannot produce enough energy.
Fumarate also helps cells repair DNA damage. This enzyme functions in the mitochondria and nucleus of cells. This means that the function of fumarate goes far beyond providing energy.
Different organisms possess different types of fumarate. Mammals possess type II fumarate. Bacteria and yeast may possess type I or type II fumarate. Yeast uses fumarate for the TCA cycle and DNA repair.
The activity of fumarate is crucial. Studies have shown that fumarate can convert up to 82% of fumarate to L-malate. Pasteurella yeast has the highest activity. The enzyme activity of cells that have undergone permeabilization treatment is four times that of normal cells.
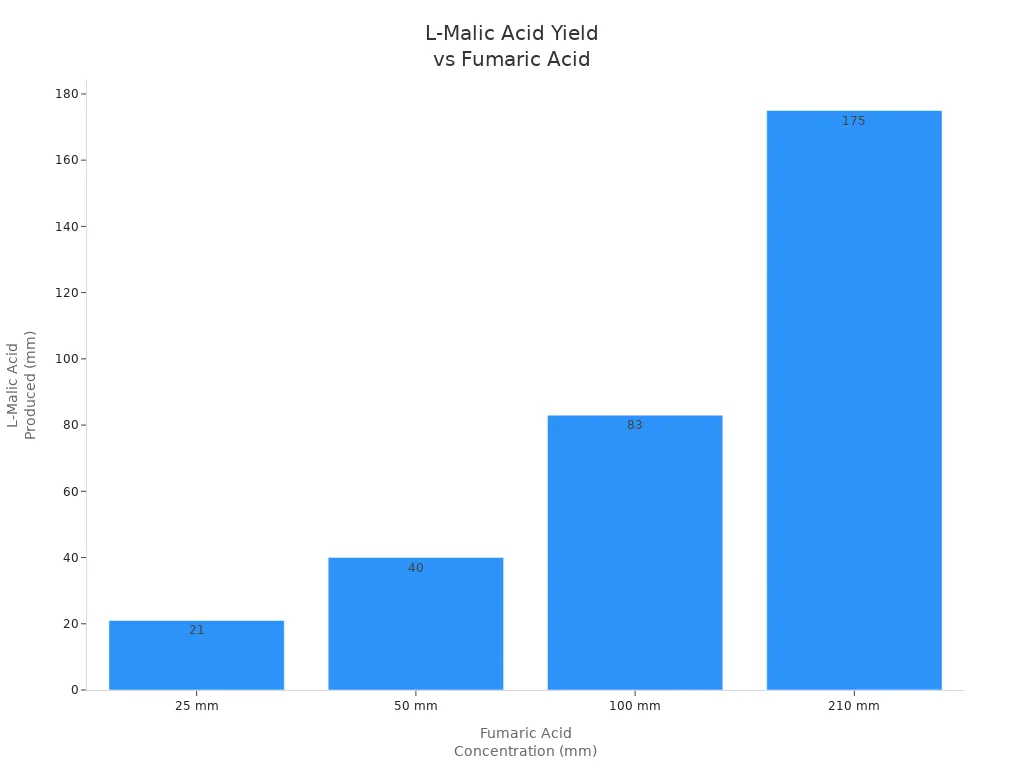
Fumarase helps the tca cycle make enough energy for cells. The enzyme links many processes and keeps cells healthy.
Fumarase converts fumarate to L-malate. This step is crucial for cellular energy and repair. This conversion is beneficial to both organisms and factories.
- L-malate is widely used in the food, beverage, pharmaceutical, and chemical industries.
- Fumarate maintains the normal functioning of the tricarboxylic acid cycle and aids in DNA repair.
| Application | Description |
|---|---|
| Food and Medicine | L-malic acid makes food taste better and helps people stay healthy. |
| Sustainable Production | Using enzymes to make L-malic acid is better for the environment. |
Fumarase helps science, factories, and nature.
FAQ
What role does fumarate enzyme play in cells?
Fumarate enzyme converts fumarate into L-malate. This step helps cells obtain energy. The enzyme functions in the mitochondria and nucleus of cells.
Where can people obtain fumarate in their daily lives?
People can obtain fumarate from food and beverages. It is also found in animal feed, skincare products, and pharmaceuticals. Fumarate sold by NORBIDAR has a wide range of uses.
Why is L-malate important for health?
L-malate helps cells produce energy. It helps repair cells and maintain metabolic stability. Scientists use L-malate to test food quality.
How does fumarate enzyme help factories and industries?
Factories use fumarate enzyme to convert fumarate into L-malate. This provides raw materials for the production of food, pharmaceuticals, and chemicals. Using enzymes is also beneficial to the environment.
Can fumarate enzyme catalyze both directions?
Fumarate enzyme can convert fumarate to L-malate and vice versa. Cells use this conversion process to convert energy and repair themselves when needed.