Fumaric acid is soluble in water, but its solubility is very low at room temperature. At 25°C, approximately 0.70 grams of fumaric acid dissolves in 100 ml of water. This means its solubility in water is lower than in some other liquids. This low solubility is crucial in food, beverage, and industrial production. It helps control the amount dissolved, thus affecting the product’s taste, texture, and stability.
Key Takeaways
- Fumaric acid has poor solubility in cold water. At room temperature, only 0.70 grams of fumaric acid dissolves in 100 ml of water.
- If the water is heated, the solubility of fumaric acid increases significantly. At 100°C, approximately 9.8 grams of fumaric acid dissolves in 100 ml of water.
- Wetting agents help fumaric acid dissolve better in water. They prevent scum formation and accelerate the dissolution process.
- Fumaric acid is widely used in food and beverages due to its sour taste. Fumaric acid is particularly suitable for use in baked goods and jellies because of its long-lasting flavor.
- Choosing the right fumaric acid is essential. You need to know its solubility in hot or cold water. This helps you achieve your desired product flavor and texture.
Is fumaric acid soluble in water?
Room temperature solubility
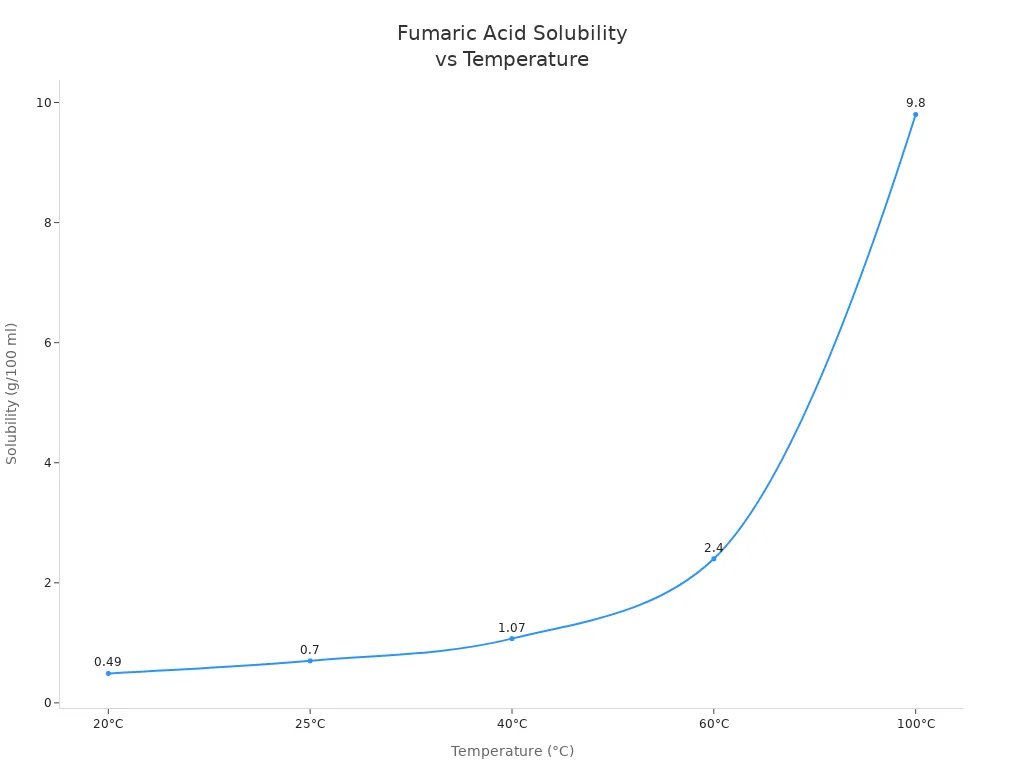
Fumaric acid is not readily soluble in water at room temperature. Scientists describe its water solubility as very poor. At 25°C, only 0.70 grams of fumaric acid dissolves in 100 ml of water. Most fumaric acid is insoluble in cold water. The table below shows the solubility at different temperatures:
| Temperature (°C) | Solubility (g/100 ml) |
|---|---|
| 20 | 0.49 |
| 25 | 0.70 |
| 40 | 1.07 |
| 60 | 2.40 |
| 100 | 9.80 |
At 25°C, approximately 6.3 grams of fumaric acid dissolves in 1 liter of water. This demonstrates that fumaric acid has very low solubility in water. Most beverages use acids with higher solubility, such as citric acid or malic acid. The low solubility of fumaric acid affects the taste and texture of beverages. Cold-water soluble fumaric acid helps address this issue. This type of fumaric acid dissolves more readily in liquids and is suitable for both food and beverages.
Note: The smaller the fumaric acid particles, the faster they dissolve. Warm water dissolves more fumaric acid than cold water.
Solubility at High Temperatures
Fumaric acid is more soluble in hot water. At 100°C, approximately 9.8 grams of fumaric acid dissolves in 100 milliliters of water. This means that hot water dissolves more fumaric acid than cold water. The following graph shows how solubility increases with increasing temperature:
Scientists have discovered that the solubility of fumaric acid increases significantly with increasing water temperature. At 189°C, its solubility is 4.84 mol/L, far exceeding its solubility at room temperature. This is crucial in factories, where heating is used to dissolve more fumaric acid before cooling and crystallizing. Similarly, maleic acid has higher solubility at higher temperatures. This indicates that acids are generally more soluble in hot water.
The solubility of fumaric acid in water depends on temperature and type. Cold-water soluble fumaric acid mixes more easily with beverages and other products. Common fumaric acid has low solubility; therefore, companies often choose other types of acids when rapid and complete dissolution is required.
Solubility Factors
The Effect of Temperature
Temperature affects the solubility of fumaric acid in water. Cold water causes fumaric acid to dissolve slowly, with only a small amount dissolving. As the water temperature rises, more fumaric acid can dissolve.
At 25°C, fumaric acid has very low solubility in water. However, its solubility increases significantly at 100°C. This is why factories heat water to promote faster and better dissolution of fumaric acid. Those working with food or chemicals need to closely monitor temperature, as it affects the solubility of fumaric acid in water.
Tip: Heating water helps dissolve fumaric acid, making it easier to use in formulations or factories.
Other Influencing Factors
Besides temperature, other factors affect the solubility of fumaric acid. The pH of the solution has an impact. If the pH is low, fumaric acid remains acidic and is difficult to dissolve. If the pH is high, it dissolves more easily.
Aqueous-alcoholic solutions also affect the solubility of fumaric acid. Scientists have found that fumaric acid behaves differently in these mixtures than in pure water. The alcohol in the mixture alters the solubility and acidity of fumaric acid. This is crucial for beverages and pharmaceuticals.
Wetting agents help fumaric acid mix with water and prevent the formation of scum on the surface. Surfactants break down fumaric acid particles, allowing them to dissolve more quickly. Glycerin is another commonly used wetting agent.
- Wetting agents help fumaric acid mix with water.
- They prevent the formation of scum on the surface.
- Surfactants break down fumaric acid particles, helping them dissolve.
These techniques are used to achieve optimal results. Appropriate temperatures, pH levels, and additives are selected to ensure fumaric acid functions best in the product.
The Importance of Fumaric Acid Solubility
Food and Beverage Applications

The solubility of fumaric acid is crucial in food and beverages. Companies test its solubility in water before use. This affects the taste, texture, and shelf life of the food. Fumaric acid imparts a rich acidity to fruits, enhancing their flavor. It helps balance sweetness and maintain pH stability. This helps inhibit the growth of harmful bacteria.
Many foods require fumaric acid to dissolve quickly. Hot water helps it dissolve faster, making it ideal for baked and cooked foods. Fumaric acid has low water absorption, so it doesn’t clump like powdered foods such as cake mix. Fumaric acid retains its activity during cooking, thus continuing to function in formulations.
NORBIDAR sells fumaric acid that is soluble in both hot and cold water. Hot water fumaric acid is suitable for baked goods and beverages. Cold water fumaric acid dissolves quickly in juices and instant beverage powders. These options help food manufacturers find the most suitable formulations.
| Application | Description |
|---|---|
| Baked Goods | Makes dough stronger, improves texture, and adds a nice sour taste in rye and sourdough. |
| Confectionery | Makes candies and jellies sour, keeps them fresh by stopping water from getting in. |
| Beverages | Keeps pH steady in juices and sodas, stops extra fermentation in wines. |
| Jams and Jellies | Makes gels stronger, lets you use less gelatin, and keeps jams fresh longer. |
Animal Nutrition & Industry
Fumaric acid is beneficial to both animals and factories. In animal feed, it mixes well with the feed, aiding digestion and growth. It also reduces the number of harmful bacteria in chickens, pigs, and fish, thereby improving feed utilization.
Fumaric acid is also used in fermentation processes in factories. Its low solubility helps increase product yield and optimize production processes. Furthermore, it reduces the generation of waste salts. NORBIDAR’s fumaric acid products are suitable for these applications and are available in both hot and cold water formulations.
| Type of Fumaric Acid | Solubility Characteristics | Applications |
|---|---|---|
| Hot Water Soluble (HWS) | Mixes quickly in hot water | Baked goods, drinks, industrial resins |
| Cold Water Soluble (CWS) | Mixes fast in cold water | Juice, instant drink powders, medicine suspensions |
The solubility of fumaric acid in water is crucial for food, animal feed, pharmaceuticals, and industrial applications. It affects product mixing methods, taste, and shelf-life properties. NORBIDAR’s fumaric acid product range allows users to choose the optimal type to meet their specific needs.
Comparison of Fumaric Acid with Other Acids

Maleic Acid
Maleic acid and fumaric acid are both dicarboxylic acids with similar chemical structures, but their solubility in water differs drastically. Maleic acid dissolves much faster than fumaric acid.
Maleic acid dissolves rapidly in water, while fumaric acid dissolves poorly, only in small amounts. This significant difference affects their applications in laboratories and factories. Maleic acid is suitable for applications requiring rapid mixing, while fumaric acid is better suited for products requiring long-term storage or slow dissolution.
At room temperature, maleic acid is almost 70 times more soluble than fumaric acid, making it a better choice for liquid mixtures.
Citric Acid
Citrate acid is another widely used acid in food and beverages. It is readily soluble in cold water, while fumaric acid is poorly soluble. The table below compares them and their uses:
| Acid | Solubility in Cold Water | Practical Uses |
|---|---|---|
| Citric Acid | Highly soluble | Good for drinks and fast flavor |
| Fumaric Acid | Low solubility | Not used much in drinks; better for solid foods with long-lasting taste |
Citric acid is commonly used in soft drinks, juices, and candies. It imparts a strong sour taste and dissolves rapidly. Fumaric acid, on the other hand, has poor solubility and is therefore often used in baked goods and jellies. It imparts a long-lasting, strong sour taste. Food manufacturers typically choose citric acid for beverages and fumaric acid for foods requiring a slower release of sourness.
Citral acid’s high solubility makes it ideal for formulations requiring rapid mixing. Fumaric acid dissolves slowly, thus preserving the rich flavor of solid foods.
Fumaric acid has poor solubility in cold water. At room temperature, only about 0.63 grams of fumaric acid dissolves in 100 ml of water. Heating the water significantly increases the amount of fumaric acid dissolved. At 100 degrees Celsius, approximately 9.8 grams of fumaric acid dissolves in 100 ml of water. Hot water accelerates molecular motion, thus dissolving fumaric acid even faster. It is necessary to monitor the amount of fumaric acid dissolved to prevent excess crystals from forming in food or pharmaceutical products.
FAQ
What does “poor solubility” mean in fumaric acid?
“Poor solubility” means that fumaric acid has very low solubility in water at room temperature. Most fumaric acid will remain solid unless the water temperature is increased.
Can people use fumaric acid in cold drinks?
Regular fumaric acid has very low solubility in cold drinks. Cold-water soluble fumaric acids like NORBIDAR have higher solubility and are suitable for instant beverages.
How to improve the solubility of fumaric acid?
Heating water helps fumaric acid dissolve. Adding a wetting agent or changing the pH also helps improve solubility.
Why do companies choose fumaric acid instead of citric acid?
Companies choose fumaric acid because it provides a long-lasting, rich sour taste and has low water absorption. It is ideal for baked goods, jellies, and foods that require a slow release of flavor.